The Occam’s Razor principle dictates that
the simplest of competing theories should be chosen. I prefer a more
colloquial variation called KISS or “Keep it Simple, Stupid.” Although
I’m a former National-Institutes-of-Health (NIH) division director, I
believe our scientific policies for generating new SCI solutions often
ignore KISS wisdom. In allegiance to the God of science and its dogma,
we have adopted complex and burdensome strategies for developing new
treatments, which - albeit ensuring the sanctity of the anointed
scientific process - create few real-world SCI treatments.
A case in point is methylprednisolone (MP), a drug
commonly administered after injury. Although NIH subjected MP to more
complex, time-consuming, and costly clinical trials than any other SCI
drug in history, many are now challenging its true effectiveness. If the
most scientifically scrutinized SCI drug can’t cut the mustard over
time, perhaps we should consider more simple options that wouldn’t take
decades to evaluate and reevaluate.
Such options include nutritional and herbal
approaches, which, given already extensive human use and presumed
safety, would be relatively easy to incorporate into treatment regimens.
For example as discussed previously, animal studies suggest that 1)
Ginkgo biloba, a widely consumed herbal medicine, 2)
melatonin,
an extensively used sleep aid, and 3) every-other-day fasting
may
be neuroprotective after injury.
This update discusses the neuroprotective potential
of two other nutritional approaches: quercetin and vitamin E.
Quercetin
Belonging to the flavonoid family of molecules,
quercetin imbues coloring to many foods, including apples, red onions,
red grapes, tomatoes, raspberries, and other berries. Evidence suggests
that it is beneficial for cancer, prostatitis, heart disease, cataracts,
allergies/inflammation, and respiratory disorders.
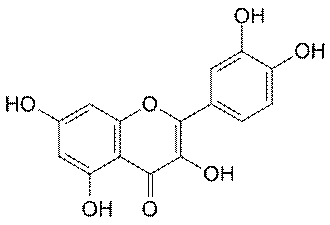
An antioxidant, quercetin scavenges free radicals
and inhibits damage-perpetuating, post-injury lipid peroxidation.
Basically, after the initial mechanical injury, a
complicated physiological chain reaction generates
free-radicals, which steal electrons from the lipids in neighboring
neuronal membranes, resulting in further cell death.
Post-injury hemorrhaging causes the
hemoglobin within red blood cells to disintegrate, releasing oxidized
iron, which triggers lipid peroxidation. Quercetin binds to this
iron, preventing it from reacting with the lipids on neighboring cells.
In addition to its antioxidant characteristics, quercetin has other
properties that augment its neuroprotective potential. For example, it
is 1) lipophilic (i.e., affinity for fat or lipid), allowing it to
diffuse through cell membranes and scavenge free radicals within the
cells, 2) anti-inflammatory, and 3) anti-edematous, i.e., inhibits
damage-causing swelling.
Dr. E. Schultke and colleagues (2003,
Canada) assessed the impact of treating experimentally injured rats with
quercetin. Different doses of quercetin or saline (i.e.,
controls) were injected into the body cavity one hour after injury and
every 12 hours thereafter for either 4 or 10 days. Recovery of hind-limb
function was evaluated by a scale which assesses functional recovery on
a gradation from 0 (no hind-limb movement) to 21 (normal walking).

Although no controls walked,
two-thirds of the quercetin-treated animals recovered some ability to do
so. Supporting the idea that iron mediates damage, the tissue of the
injured cords of control animals tested positive for iron, but no iron
was detected in the cords of quercetin-treated animals.
The investigators reported the
results of a somewhat similar investigation in 2009. Although no control
animals regained sufficient hind-limb function to walk, approximately
50% of the rats treated with twice-daily doses of quercetin over three
or ten days were able to walk. In general, the rats that were treated
with quercetin for a longer duration recovered more function. Compared
to controls, more spinal-cord tissue was preserved in the
quercetin-treated rats.
Vitamin E
Vitamin E is a generic term for a class of
molecules called tocopherols, the most physiologically ubiquitous being
alpha-tocopherol (illustration). Vitamin E is found in vegetable oils,
whole grains, dark green leafy vegetables, nuts and seeds, and legumes.
Supplementation may promote cardiovascular and eye health, and prevent
cancer and age-related cognitive decline.
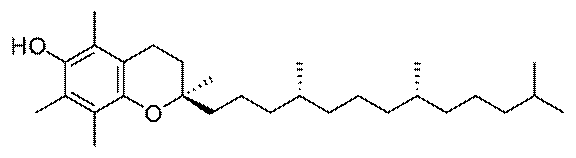
Like quercetin, vitamin E is an antioxidant that
protects neuronal membranes from
lipid-peroxidation.
Dr. Royal Saunders
and co-investigators (1987, Ohio) investigated the effects on lipid
peroxidation of treating experimentally injured cats with a combination
of vitamin E and another antioxidant, selenium. Cats were orally
pretreated with the combination for five days before injury. Compared
to untreated controls, the spinal-cord tissue at the injury site of the
antioxidant-treated cats had fewer molecules associated with
lipid-peroxidation.
Former PVA scientific advisor,
Dr. Douglas Anderson et al (1988, Ohio) evaluated vitamin-E’s effect
in cats with experimental SCI. The cats were treated orally with the
vitamin for five days before and after injury. Functional recovery was
assessed by improvements in walking, running, and stair climbing.
Four weeks post-injury, vitamin-E
treated cats recovered 72% of their pre-injury function compared to only
20% for similarly injured but untreated controls. The investigators
concluded that
 vitamin-E
pretreatment “was extraordinarily effective in promoting functional
recovery…” However, they noted that because vitamin E enters the central
nervous system slowly, it’s probably not a viable candidate for treating
SCI. (see below)
vitamin-E
pretreatment “was extraordinarily effective in promoting functional
recovery…” However, they noted that because vitamin E enters the central
nervous system slowly, it’s probably not a viable candidate for treating
SCI. (see below)
Dr. Kenichi Iwasa
and associates (1989, Japan) compared the recovery of rats fed a diet
containing vitamin E at a level 25 times that fed to controls. The high
vitamin-E diet was consumed for eight to ten weeks before experimental
injury. Hind-limb function was assessed using a scale ranging from 0 (no
movement) to 4 (complete recovery). One day after injury, the
vitamin-E-treated rats had a hind-limb score of 3.5 compared to 2.4 for
controls. In addition, vitamin-E supplementation enhanced the recovery
of nerve conductivity and spinal-cord blood flow, and reduced the level
of lipid-peroxidation-associated molecules. Finally, microscopic
examination of the injured cord tissue showed less damage, such as
bleeding and edema, in vitamin-E-treated animals.
This investigative team later
(1990) compared recovery in rats fed the aforementioned control diet
with rats fed a vitamin-E-deficient diet (specifically, 20-times
less). In other words, this study compared controls to
vitamin-E-deficient and not -supplemented animals. The results indicated
that vitamin-E-deficient rats had 1) less recovery of hind-limb
function, 2) less restoration of spinal-cord blood flow, 3) more
compromised nerve conduction, 4) more bleeding and edema, and 5) a
greater production of lipid-peroxidation-related chemicals.
Recently, Dr. Al Jadid and colleagues (2009,
Saudi Arabia) reconfirmed vitamin E’s neuroprotective effects. Injured
rats were divided into three groups: saline-treated controls and two
groups that received different levels of vitamin E. Supplementation was
started at the time of injury and continued for 14 days.
Post-injury functional recovery was
measured using an activity cage, which uses horizontal and vertical
sensors to measure movements. Rats who recovered greater function would
trigger the sensors more. Both vitamin-E supplemented groups had
statistically significant functional improvements by the end of the
study compared to controls. The results suggest that vitamin E, even
when administered after injury, may be a useful SCI treatment option.
Conclusion
Clearly, many therapeutic agents look promising in
animal research, but fall short when used in humans. Although I have no
idea whether quercetin, vitamin E or other nutritional approaches are
truly neuroprotective after human injury, common sense suggests
that there is little to lose and perhaps much to gain by including them
in our SCI-treatment regimen.
Adapted from article appearing in February 2010 Paraplegia News (For subscriptions,
call 602-224-0500) or go to
www.pn-magazine.com.
TOP